- Home
- Types & Treatments
- Myeloma
- Treatments
Myeloma
Myeloma Treatment
At The University of Kansas Cancer Center, you have access to the latest therapies and treatment options for multiple myeloma such as chemotherapy and radiation therapy. You will receive care from an experienced team of medical oncologists, hematologists, nurse practitioners and other medical professionals. The goals of multiple myeloma treatment are:
- Reduce the multiple myeloma symptoms you may be experiencing.
- Control progression of the disease.
- Provide long-term remission.
You and your oncologist will discuss any therapy you may require for your multiple myeloma treatment plan. Depending on your multiple myeloma diagnosis, you may receive therapies alone or in combination.
Myeloma treatment options

More options, more hope
Many new therapies are available through clinical trials. Find out how you may benefit from a clinical trial, and what it can do for others.
Life after myeloma
Surviving multiple myeloma is a lifelong process that requires regular and ongoing checkups. You also may need help managing some of the side effects of your myeloma treatment. Your healthcare team can let you know about any lifestyle changes you can make to help speed recovery and improve your overall quality of life. You also may wish to visit the Brandmeyer Patient Resource Center for more information.
Our experienced team of doctors, nurses, counselors, dietitians and research coordinators can help you:
- Understand your treatment options.
- Assess and manage side effects from your treatments.
- Monitor for cancer recurrence.
- Manage complications from your condition.
- Connect you with relevant clinical studies.
- Make recommendations for lifestyle changes.
- Keep an eye on your overall health.
Helpful websites to learn more about myeloma:
- International Myeloma Foundation
- Leukemia & Lymphoma Society
- National Marrow Donor Program
- National Cancer Institute (part of the U.S. National Institutes of Health)
- American Cancer Society
The University of Kansas Cancer Center does not assume responsibility for any of the information posted on these sites.
Related links
Start your path today.
Your journey to health starts here. Call 913-588-1227 or request an appointment at The University of Kansas Cancer Center.
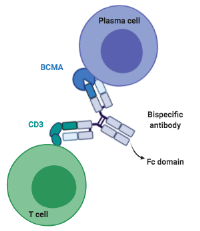 Bispecific T-cell engagers are a form of immunotherapy that use the patient’s own immune cells, the T cells, to engage and destroy the cancer cells. A bispecific antibody can be described as a 2-forked molecule that forms a bridge between cancer cells and T cells. One fork attaches to the T cell and the other fork attaches to the cancer cell. This allows the T cell to get close to the cancer cell, punch a hole in it and destroy it. Bispecific T-cell engager immunotherapies that we offer and are approved by the US Food and Drug Administration to treat multiple myeloma include:
Bispecific T-cell engagers are a form of immunotherapy that use the patient’s own immune cells, the T cells, to engage and destroy the cancer cells. A bispecific antibody can be described as a 2-forked molecule that forms a bridge between cancer cells and T cells. One fork attaches to the T cell and the other fork attaches to the cancer cell. This allows the T cell to get close to the cancer cell, punch a hole in it and destroy it. Bispecific T-cell engager immunotherapies that we offer and are approved by the US Food and Drug Administration to treat multiple myeloma include: